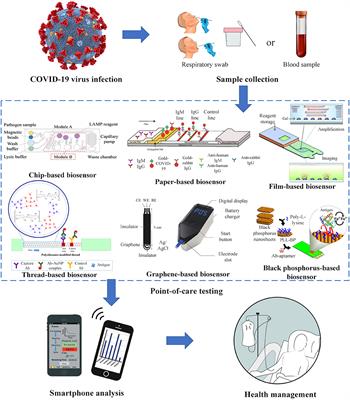
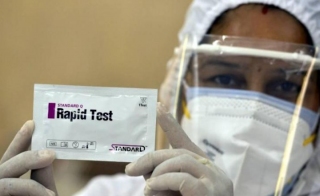
S D Biosensor Covid Antibody Test

Seven of them are chinese companies.
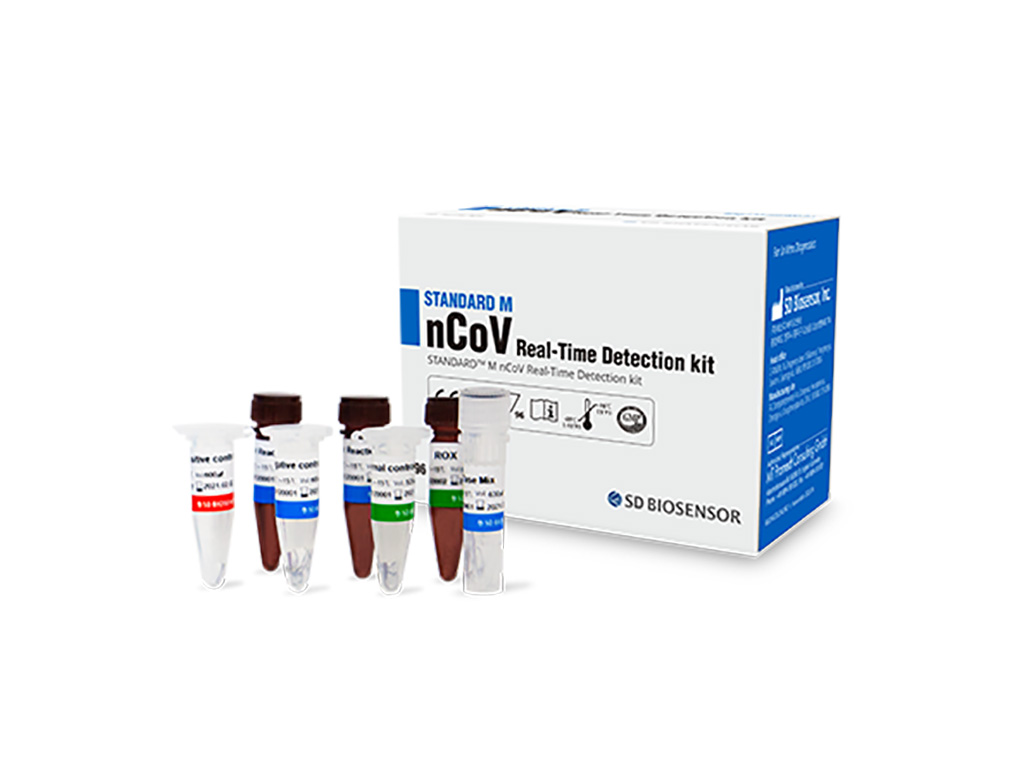
S d biosensor covid antibody test. Hsic on thursday announced the availability of an antibody rapid blood test known as standard q covid 19 igm igg rapid test intended to be administered at the. The standard q covid 19 test delivers results in minutes credit. Announced on march 26 2020 the availability of an antibody rapid blood test known as standard q covid 19 igm igg rapid test. Just 10ul of specimen.
The standard q covid 19 test is a rapid immunochromatography test designed for the qualitative presumptive detection of specific igm and igg antibodies associated with the novel coronavirus sars cov 2 in blood drawn with a pinprick. Sd biosensor standard f covid 19 igm igg combo fia biomedomics covid 19 igm igg rapid test the investigation of the antibody response to sars cov 2 in a laboratory setting may involve the measurement of igg igm and iga antibodies as well as the specific targets of these antibodies. Overall 16 companies can now market their test kits in the country. Rapid testing for sars cov 2 antibodies within 10 minutes.
In india the icmr has allowed the use of antigen detection kits developed by the south korean company s d biosensor which has a manufacturing unit in manesar. The government also approved 12 rapid antibody test kits for covid 19 diagnosis. Standard q covid 19 igm igg duo test kit is a rapid immunochromatography test designed for the qualitative presumptive detection of specific igm and igg to sars cov 2 in humoral fluid. Standard q covid 19 igm igg duo test kit is a rapid immunochromatography test designed for the qualitative presumptive detection of specific igm and igg to sars cov 2 in humoral fluid.
Suitable for point of care testing. Whole blood serum plasma. The kit commercially called standard q covid 19 ag detection kit comes with a with an inbuilt covid antigen test device viral extraction tube with viral lysis buffer and sterile swab for sample collection. Rttnews henry schein inc.
This product performs qualitative analysis by detecting influenza virus a type and b type antigens in the nasopharyngeal sample.