S D Biosensor Covid Antigen Test

Standard q covid 19.
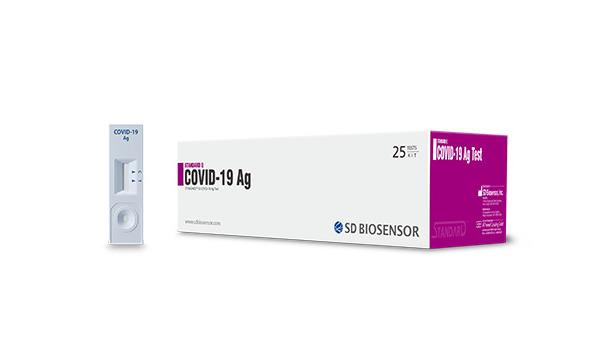
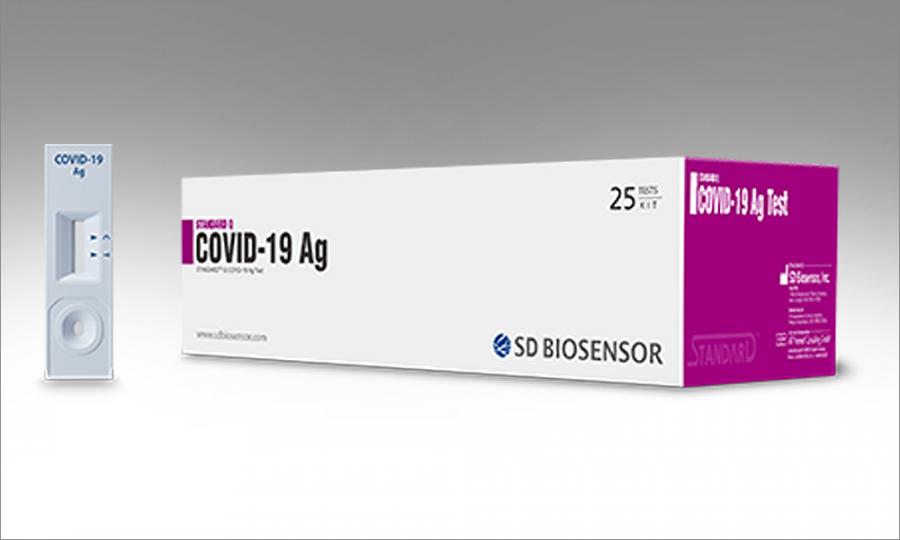
S d biosensor covid antigen test. Bgms sd check gold sd codefree codefree plus codefree plus nfc codefree plus bt mentor mentor nfc mentor bt gluconavii gdh gluconavii nfc gluconavii pro. The kit commercially called standard q covid 19 ag detection kit comes with a with an inbuilt covid antigen test device viral extraction tube with viral lysis buffer and sterile swab for sample collection. Standard q covid 19 ag test. Be performed to demonstrate that a covid 19 test for home non.
The antigen test approved by icmr is a point of care test performed outside the conventional laboratory setting and is used to quickly obtain the results. Since founded we have been establishing a successful foothold in order to provide worldwide blood glucose monitors. A 5 diagnostic test for covid 19 won emergency authorization from the us food and drug. And standard q from south korea s sd biosensor.
Sd biosensor is represented by velocitydx in the united states. About us sd biosensor our history our vision. In india the icmr has allowed the use of antigen detection kits developed by the south korean company s d biosensor which has a manufacturing unit in manesar. Henry schein intends to add other tests from velocitydx and other suppliers specific to covid 19 with the goal of offering a wide array of point of care tests for health care professionals.
Once collected the swab needs to be directly put into the extraction buffer in tube provided in the kit which inactivates the virus. This product performs qualitative analysis by detecting influenza virus a type and b type antigens in the nasopharyngeal sample. Standard q covid 19 ag test is a rapid chromatographic immunoassay for the qualitative detection of specific antigens to sars cov 2 present in human nasopharynx. Roche s test is a rapid chromatographic immunoassay intended for the qualitative.
Standard q covid 19 ag detection kit developed by sd biosensor a south korea based company is approved by icmr. As the covid 19 pandemic continues into the fall roche group is planning to launch its latest tool later this month. Sd biosensor site map. The standard q covid 19 test is manufactured by sd biosensor inc a global bio diagnostic company.
Tests on individuals with negative antigen test or other point of care test. And while its sars cov 2 rapid antigen test will be available in markets accepting the ce mark the company is expecting the filing for emergency use authorization eua from the u s.