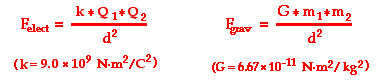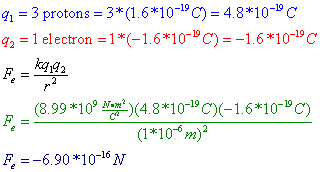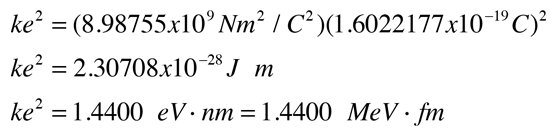Value K Physics Coulomb S Law

Where ε 0 is known as the permittivity of free space.
Value k physics coulomb s law. In si units it is equal to 8 987 551 7923 14 10 9 kg m 3 s 2 c 2. F k q 1 q 2 r 2 where k is a constant which has a value of 9 10 9 nm 2 c 2 r distance in meter m q 1 q 2 charges expressed in coulomb c f electrical force in newton n suppose we consider vacuum as the medium between the two poles then constant k is replaced by 1 4 π ε 0 where ε 0 is electrical permittivity of. Thus we have k 1 4 π. Permittivity is a property of a material that is indicative of how well it supports an electric field but is beyond the scope of these notes.
Asked on december 27 2019 by gurram shaikh. If the charged objects are present in water the value of k can be reduced by as much as a. Charles agustin coulomb 1736 1806 measured electrical attractions and repulsion quantitatively and deduced the law that governs them. The symbol k is a proportionality constant known as the coulomb s law constant.
And yes the cgs unit system sets it equal to 1 without losing any usefulness of the equation. The value of the constant k in coulomb s law is. Coulomb s law states that force of attraction or repulsion between two electric charges is directly proportional to their magnitudes and inversely proportional to the square of the distance between them coulomb s law examples are given below. The quantity of electrostatic force between stationary charges is always described by coulomb s law.
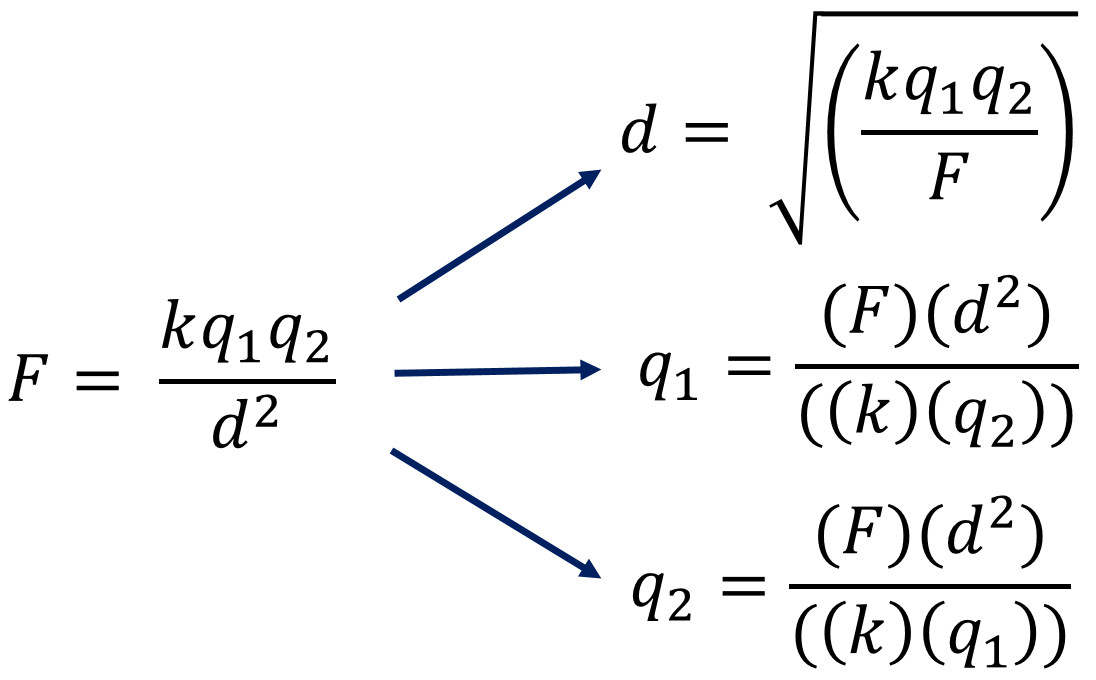
The force between two point charges is directly proportional to the magnitude of each charge q 1 q 2 inversely proportional to square of the separation between their centers r directed along the separation vector connecting their centers r this relationship is known as coulomb s law. The value of this constant is dependent upon the medium that the charged objects are immersed in. F q 1 q 2 4 π ε 0 r 2. The electric force between charged bodies at rest is conventionally called electrostatic force or coulomb force.
More traditionally coulomb s law is written. And the purpose of coulomb s law coulomb s law is to predict what is going to be the force of the electrostatic force of attraction or repulsion between two forces. The coulomb constant the electric force constant or the electrostatic constant denoted k e k or k is a proportionality constant in electrostatics equations. Coulomb s law or coulomb s inverse square law is an experimental law of physics that quantifies the amount of force between two stationary electrically charged particles.
Charles augustin coulomb 1736 1806 france. ε 0 8 85 10 12 f m 1 farads per metre. Electric charges and fields. It was named after the french physicist charles augustin de coulomb 1736 1806 who introduced coulomb s law.
However in the mks system of units every ot. In the case of air the value is approximately 9 0 x 10 9 n m 2 c 2.